For%20the%20cyclic%20process%20shown,%20W%20is:D - PowerPoint PPT Presentation
Title:
For%20the%20cyclic%20process%20shown,%20W%20is:D
Description:
For the cyclic process shown, Q is:D ... cannot determine We saw that there is always heat rejected in a thermodynamic cycle ... How much work is done by the ... – PowerPoint PPT presentation
Number of Views:128
Avg rating:3.0/5.0
Title: For%20the%20cyclic%20process%20shown,%20W%20is:D
1
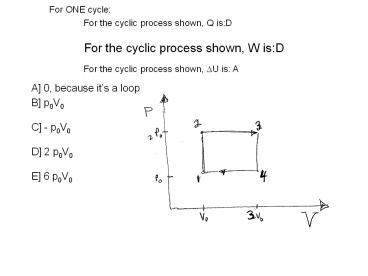
For the cyclic process shown, W isD
For ONE cycle
For the cyclic process shown, Q isD
For the cyclic process shown, ?U is A
- A 0, because its a loop
- B p0V0
- C - p0V0
- D 2 p0V0
- E 6 p0V0
2
Is ALL heat that we add converted into work?In
other words, is there any part of the cycle where
heat is removed from the gas?
- A No heat is removed
- B Segment 3-4 only
- C Segment 4-1 only
- D Segments 3-4-1
- E Segments 3-4-1-2
3
An ADIABATIC PROCESS is one in which no heat
transfer occurs. Q0.If we expand a gas from VA
to VB adiabatically, what will be the final
pressure?
- A More than PB
- B Less than PB
- C Equal to PB
- D cannot determine
4
We saw that there is always heat rejected in a
thermodynamic cycle Qc
5
Otto cycle -- in your Ottomobile.
http//www.youtube.com/watch?vE0PIdWdw15U
6
THIS SIDE OF THE CYCLE IS ACTUALLY AN ENTIRE
COMPRESSION EXPANSION TO TAKE IN NEW AIR/FUEL.
7
How much work is done by the gas in the cycle
shown? D
How much total heat is added to the gas in the
cycle shown? D
- A 0
- B p0V0
- C 2p0V0
- D -2p0V0
- E 4 p0V0
If negative heat is added to the gas, this
means more heat is expelled from the gas than
taken in. (The difference is the work done on the
gas.)
8
In one (ccw) Carnot cycle shown, the work done by
the gas is
- A
- B -
- C 0
9
Work lt 0, so Q lt 0. Along which paths is heat
expelled from the gas?
- A 1-2
- B 2-3
- C 3-4
- D 4-1
- E none
10
Heat is expelled from the gas during isothermal
compression 3-4. Heat is added to the gas during
isothermal expansion 1-2. More heat is expelled
than added. The net effect is to take heat from
a cold reservoir, and add it to a hot reservoir
(along with some extra heat from the work done on
the gas.) This is a fridge!
11
Monday 9/13The laws of mechanics (and EM, etc.)
are time-reversal invariant. So how come this
looks funny?
- http//www.youtube.com/watch?vmGZjCUKowIs
12
Heat flows from a hot object to a cold object in
contact with it becauseA the hot object has
more total internal energy -and heat flows until
both objects have the same internal energyB
the hot object has more total energy per
molecule- and heat flows until both objects have
the same energy per moleculeC the hot object
has more translational kinetic energy per
molecule - and heat flows until both objects have
the same translational kinetic energy per
molecule.
13
Ideal, monatomic gas goes around the cycle
shown.Is this an engine or a fridge?
A engine B fridge
14
What is the temperature at c?
A Ta B 2Ta C 3Ta D Ta/3 E cannot
determine
15
TbTa.
So Tc 3Ta
How much work does the gas do a-b? Use paper
pencil
A p0V0 B - (2/3) p0V0 C - p0V0 ln(3) D -
p0V0 ln(1/3) E cannot determine
16
Tc 3Ta
Wab - p0V0 ln(3)
What is the pressure at b?
A p0 B 2p0 C -2p0 D 3p0 E cannot
determine
17
Tc 3Ta
Wab - p0V0 ln(3)
Pb 3P0
What is the work done by the gas b-c?
A p0V0 B 3 p0 (1/3) V0 C 3 p0 (2/3)
V0 D 0 E cannot determine
18
Tc 3Ta
Wab - p0V0 ln(3)
Pb 3P0
Wbc 3 p0 (2/3) V0
What is the work done by the gas c-a?
A p0V0 B 3 p0 (1/3) V0 C 3 p0 (2/3)
V0 D 0 E cannot determine
19
Tc 3Ta
Wab - p0V0 ln(3)
Wbc 2 p0V0 Wca 0
Pb 3P0
Along which segments is heat added?
A ab B bc C ca D ab bc E bc ca
20
Tc 3Ta
Wab - p0V0 ln(3)
Wbc 2 p0V0 Wca 0
Pb 3P0
Heat is added only along bc. How much heat is
added?
A 2p0V0 B nCvTa C nCpTa D nCp 2Ta E nCp
3Ta
21
Tc 3Ta
Wab - p0V0 ln(3)
Qbc nCp 2Ta
Wbc 2 p0V0 Wca 0
Pb 3P0
Now p0V0 nRTa. Lets find the efficiency!
e W/Qadded
22
A cylinder containing an ideal gas is heated at
constant pressure from 300K to 350K by immersion
in a bath of hot water. Is this process
reversible or irreversible?
A reversible B irreversible
23
A hot piece of metal is placed in an insulating
box filled with a polyatomic gas.When thermal
equilibrium has been reached
- A the metal and the gas have equal total energy
- B the average energy per atom in the metal is
equal to the average energy per molecule in the
gas - C the average kinetic energy per atom in the
metal is equal to the average translational
kinetic energy per molecule in the gas - D the average kinetic energy per atom in the
metal is equal to the average kinetic energy per
atom in the gas
24
W 9/15What is the work done by the gas in the
reversible isothermal expansion shown?
A p0V0ln(2) B p0V0 C 2 p0V0 D 0 E none
of these
What is the heat added, Q?
A
25
No change in internal energy, so WQ
p0V0ln(2).What is the entropy change of the gas?
A p0V0ln(2) B nRln(2) C nRln(1/2) D 0 E
cannot determine
?S Q/T for an isothermal process. Use
p0V0nRT along with Q p0V0ln(2) to find ?S
nRln(2).
What is the entropy change in the hot reservoir
which isadding heat to the gas?
26
In a reversible process, ?S 0. So the entropy
change in the hot reservoir (which is at the same
temperature T as the gas) is -nRln(2). Answer C.
What is the entropy change in the hot reservoir
which isadding heat to the gas?
A p0V0ln(2) B nRln(2) C nRln(1/2) D 0 E
cannot determine
27
We showed, for a Carnot cycle, that QH/TH
Qc/TC -Qc/TcWhat is the change in entropy of
the gas around the entire Carnot cycle?
A p0V0ln(2) B nRln(2) C nRln(1/2) D 0 E
cannot determine
28
Any reversible process consists of adjoining
Carnot cycles. ?S for adjoining segments cancels.
So Entropy, like Internal Energy, is a state
variable, and depends only on the state of a
system (p, V for a gas). -gt You can calculate
entropy changes for irreversible processes by
taking a reversible path to the same endpoint.
Example free expansion to double the volume.
Tf Ti.
29
Entropy changes in non-isothermal processes
Example 1 heating water Example 2a/b heating a
gas at constant V/p