Coloring the Periodic Table Families PowerPoint PPT Presentation
1 / 40
Title: Coloring the Periodic Table Families
1
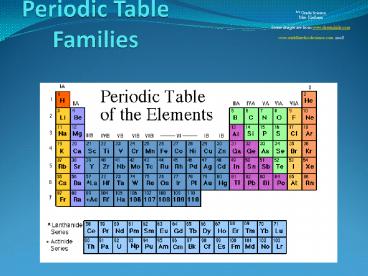
Coloring the Periodic Table Families
- 8th Grade Science
- Mrs. Kielbasa
- Some images are from www.chem4kids.com
- www.middleschoolscience.com 2008
2
(No Transcript)
3
Why is the Periodic Table important to me?
- The periodic table is the most useful tool to a
chemist. - You get to use it on every test.
- It organizes lots of information about all the
known elements.
4
Pre-Periodic Table Chemistry
- was a mess!!!
- No organization of elements.
- Imagine going to a grocery store with no
organization!! - Difficult to find information.
- Chemistry didnt make sense.
5
Dmitri Mendeleev Father of the Table
- HOW HIS WORKED
- Put elements in rows by increasing atomic weight.
- Put elements in columns by the way they reacted.
- SOME PROBLEMS
- He left blank spaces for what he said were
undiscovered elements. (Turned out he was
right!) - He broke the pattern of increasing atomic weight
to keep similar reacting elements together.
6
The Current Periodic Table
- Mendeleev wasnt too far off.
- Now the elements are put in rows by increasing
ATOMIC NUMBER!! - The horizontal rows are called periods and are
labeled from 1 to 7. - The vertical columns are called groups are
labeled from 1 to 18.
7
GroupsHeres Where the Periodic Table Gets
Useful!!
- Elements in the same group have similar chemical
and physical properties!! - (Mendeleev did that on purpose.)
- Why??
- They have the same number of valence electrons.
- They will form the same kinds of ions.
8
Families on the Periodic Table
- Elements on the periodic table can be grouped
into families bases on their chemical properties. - Each family has a specific name to differentiate
it from the other families in the periodic table. - Elements in each family react
- differently with other elements.
- Families may be one column,
- or several put together.
- Families have names rather
- than numbers. (Just like your
- family has a common last name.)
9
Hydrogen
- Hydrogen belongs to a family of its own.
- Hydrogen is a diatomic, reactive gas.
- Hydrogen was involved in the explosion of the
Hindenberg. - Hydrogen is promising as an alternative fuel
source for automobiles
10
Hydrogen is a non-metal
1 Electron
11
Alkali Metals
- 1st column on the periodic table (Group 1) not
including hydrogen. - Very reactive metals, always combined with
something else in nature (like in salt). - Soft enough to cut with a butter knife
12
ALKALI METALS
- Group 1
- Hydrogen is not a member, it is a non-metal
- 1 electron in the outer shell
- Soft and silvery metals
- Very reactive, esp. with water
- Conduct electricity
Image http//www.learner.org/interactives/periodi
c/groups2.html
13
Alkaline Earth Metals
- Second column on the periodic table. (Group 2)
- Reactive metals that are always combined with
nonmetals in nature. - Several of these elements are important mineral
nutrients (such as Mg and Ca)
14
ALKALINE EARTH METALS
- Group 2
- 2 electrons in the outer shell
- White and malleable
- Reactive, but less than Alkali metals
- Conduct electricity
15
Transition Metals
- Elements in groups 3-12
- Less reactive harder metals
- Includes metals used in jewelry and construction.
- Metals used as metal.
16
TRANSITION METALS
- Groups in the middle (3-12)
- Good conductors of heat and electricity.
- Some are used for jewelry.
- The transition metals are able to put up to 32
electrons in their second to last shell. - Can bond with many elements in a variety of
shapes.
17
Boron Family
- Elements in group 13
- Aluminum metal was once rare and expensive, not a
disposable metal.
18
BORON FAMILY
- Group 13
- 3 electrons in the outer shell
- Most are metals
- Boron is a metalloid
19
Carbon Family
- Elements in group 14
- Contains elements important to life and
computers. - Carbon is the basis for an entire branch of
chemistry. - Silicon and Germanium are important
semiconductors.
20
CARBON FAMILY
- Group 14
- 4 electrons in the outer shell
- Contains metals, metalloids, and a non-metal
Carbon (C)
21
Nitrogen Family
- Elements in group 15
- Nitrogen makes up over ¾ of the atmosphere.
- Nitrogen and phosphorus are both important in
living things. - Most of the worlds nitrogen is not available to
living things. - The red stuff on the tip of matches is phosphorus.
22
NITROGEN FAMILY
- Group 15
- 5 electrons in the outer shell
- Can share electrons to form compounds
- Contains metals, metalloids, and non-metals
23
Oxygen Family or Chalcogens
- Elements in group 16
- Oxygen is necessary for respiration.
- Many things that stink, contain sulfur (rotten
eggs, garlic, skunks,etc.)
24
OXYGEN FAMILY
- Group 16
- 6 electrons in the outer shell
- Contains metals, metalloids, and non-metals
- Reactive
25
Halogens
- Elements in group 17
- Very reactive, volatile, diatomic, nonmetals
- Always found combined with other element in
nature . - Used as disinfectants and to strengthen teeth.
26
Halogens
- Group 17
- 7 electrons in the outer shell
- All are non-metals
- Very reactive are often bonded with elements from
Group 1
27
The Noble Gases
28
The Noble Gases
- Elements in group 18
- VERY unreactive, monatomic gases
- Used in lighted neon signs
- Used in blimps to fix the Hindenberg problem.
- Have a full valence shell.
29
Noble Gases
- Group 18
- Exist as gases
- Non-metals
- 8 electrons in the outer shell Full
- Helium (He) has only 2 electrons in the outer
shell Full - Not reactive with other elements
30
Lanthanide - Rare Earth Metals
- Some are radioactive
- silver, silvery-white, or gray metals.
- Conduct electricity
- Burn easily in air
- High melting points and boiling points.
31
Actinides -
- All are radioactive.
- highly electropositive.
- metals tarnish readily in air.
- very dense metals with distinctive structures.
- react with boiling water or dilute acid to
release hydrogen gas. - combine directly with most nonmetals.
32
(No Transcript)
33
(No Transcript)
34
(No Transcript)
35
(No Transcript)
36
Periodic Table Song
- https//www.youtube.com/watch?v-I7l8TgtuLQ
- https//www.youtube.com/watch?vDYW50F42ss8
37
(No Transcript)
38
(No Transcript)
39
(No Transcript)
40
(No Transcript)