Chemical Kinetics : rate of a chemical reaction - PowerPoint PPT Presentation
Title:
Chemical Kinetics : rate of a chemical reaction
Description:
Before a chemical reaction can take place the molecules involved must be raised to a state of higher potential energy. They are then said to be activated or to form ... – PowerPoint PPT presentation
Number of Views:585
Avg rating:3.0/5.0
Title: Chemical Kinetics : rate of a chemical reaction
1
- Chemical Kinetics rate of a chemical reaction
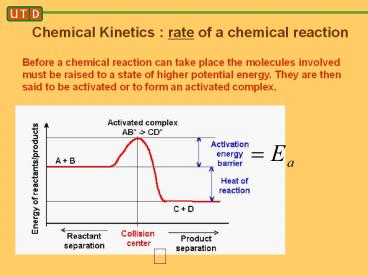
Before a chemical reaction can take place the
molecules involved must be raised to a state of
higher potential energy. They are then said to be
activated or to form an activated complex.
2
- In 1889 Arrhenius said
- vant Hoff eq. for temperature coefficient of
equilibrium - constant is
2) mass-action law relates equilibrium
constant to the ratio of rate constants
Hence a reasonable eq. for the variation of rate
constant with temperature is
Where Ea is the activation energy of the reaction
3
- If Ea does not depend on temperature, we can
integrate this last eq. to obtain
where ln A is the constant of integration. Hence
This is the famous Arrhenius eq. for the rate
constant. According to Arrhenius, molecules must
acquire a certain critical energy Ea before they
can react. The Boltzmann factor
is the fraction of molecules that manages to
obtain the necessary energy. This interpretation
is still held to be essentially correct.
4
- Henry Eyring (1901-1981)
The rate of any chemical reaction can be
formulated in terms of its activated complex.
The rate of reaction is the number of activated
complexes passing per second over the top of the
potential energy barrier. This rate is equal to
the concentration of activated complexes times
the average velocity with which a complex moves
across to the product side.
Calculation of conc. of activated complexes is
greatly simplified if we assume that they are in
equil. with the reactants. This equil. can then
be treated by means of thermodynamics or
statistical mechanics.
5
- Transition State Theory
Consider this equilibrium
equil. constant for the formation of the complex
is
the conc. of complexes is thus
according to transition state theory, the rate of
reaction is
The rate of passage over the barrier is equal to
the frequency with which the complex flies apart
into the products.
6
- The complex flies apart when one of its
vibrations becomes a translation, and what was
formerly one of the bonds holding the complex
together becomes simply the line of centers
between separating fragments.
The frequency is equal to where
is the average energy of the vibration leading
to decomposition. Since by hypothesis this is a
thoroughly excited vibration at temperature T, it
has its classical energy and hence
frequency
The reaction rate is therefore
with rate constant
7
- This is the general expression given by
transition state theory for the rate constant of
any elementary reaction. To be precise, the
expression for k2 should be multiplied by a
factor called the transmission coefficient,
which is the probability that the complex will
not recross the transition state and dissociate
back into products. In basic TST,
The activated complex is similar to a normal
stable molecule in every respect save one. The
sole difference is that one of its vibrational
degrees of freedom is missing, having been
transformed into the translation along the
reaction coordinate. Instead of 3N-6 vibrational
modes, it has 3N-7 modes (non-linear case).
8
- We can formulate k2 in thermodynamic terms by
introducing the standard free energy change
This is the difference between the free energy of
the activated complex and that of the reactants,
when all are in their standard states.
The quantities
are called the free energy of activation, the
heat of activation, and the entropy of
activation. The heat of activation is almost
equivalent to the experimental energy of
activation Ea, except for a PV term which is
negligible for solid or liquid systems.
9
- Chemical
Before
can correct for a wrong choice of transition
state this way as well
10
- challenges for computational modeling
- where/what is the transition state?
- what is the reaction coordinate?
Schematic representation of the free energy
landscape with two stable, attractive wells
separated by a transition state ridge, which
connects the highest free energy points of all
possible paths connecting the reactant and
product states. The dotted line represents a new
trajectory that was branched of at point p from
an old trajectory (bold line) and surpasses the
TS ridge at a lower point.
11
- Chemical
12
- Chemical
Before
13
- Chemical
Before
14
- Chemical
Before
15
- Chemical
Before
16
- Chemical
Before
17
- Chemical
Before
18
- Example of a complicated reaction coordinate
aqueous proton transfer reaction
what is the reaction coordinate?
19
- Chemical
Before
20
- Transition path sampling
21
- Chemical
22
- Chemical
23
- Chemical