Adsorption PowerPoint PPT Presentation
Title: Adsorption
1
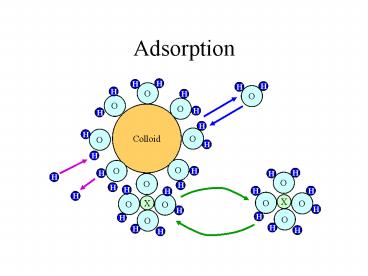
Adsorption
H
H
H
H
O
O
H
H
O
O
Colloid
H
H
H
O
O
H
H
H
O
O
H
H
H
H
O
H
O
H
H
H
H
H
H
H
O
O
O
O
X
X
H
H
O
O
H
H
H
H
H
H
2
Types of Adsorption
?S(H2O)m X(H2O)nq ? ? SX(H2O)pr (q-r)H
(mn-p)H2O
- Physical adsorption - van der Waals forces
- Electrostatic adsorption - electric charge
- Chemical adsorption - bond to solid
3
Linear Distribution Coefficient
4
Freundlich Isotherm
5
Langmuir Isotherm
6
Comparison of Isotherms
Kd 1.5 Freundlich K 1.5, n 0.9 Langmuir K
1.5, Cmax 30
7
Acid-Base Equilibria
- Dissociate surface
- ?S?OH H ? ?S?OH2
- Protonate surface
- ?S?OH ? ?S?O- H
- pH where the net surface charge is zero is the
zero point of charge ZPC
8
ZPC
?S?OH2 ? ?S?OH H
?S?OH ? ?S?O- H
9
Surface Titration
At any point of the titration the Proton
condition is given by
The net number of moles of bound OH- is given by
The mean charge is given by
10
Surface Titration
If we know the surface area S
The mean charge is given by
If ions other than H are absorped then
11
ZPC - Isoelectric Point
ve surface charge
-ve surface charge
Cback elec 0.001 0.003 0.01 0.03
pHZPC
CB
CA
12
Typical Isoelectric Points
13
Specific Adsorption
H
H
Non-specific
Substrate
O
H
H
O
O
Outer Shell
H
H
H
H
O
H
H
H
O
H
O
O
O
H
H
O
H
H
O
O
H
H
Inner Shell
H
H
O
H
H
Specific
H
H
14
Cation and Anion Adsorption
100
adsorbed
Cations
Anions
0
pH
Sorption edge
15
Electrostatic Adsorption Models
- Sorbing surface contains functional groups that
react with sorbing solutes to form surface
complexes - Surface complexing can be described my
mass-action equations (K) corrected for
electrostatic effects
16
Electrostatic Adsorption Models
- Surface charge ? and electrical potential ? are
consequences of chemical reactions involving the
surface groups - Binding constants are empirical parameters
related to the intrinsic thermodynamic constants
by the activity coefficient of the surface
species.
17
Constant Capacitance Model
Counter ions
?1
Distance ?
OH O - OH OH2 OM L OH
?0
?0 ?
18
Diffuse Double- Layer Model
-
?d
-
-
Distance ?
OH O - OH OH2 OM L OH
?d
-
?0
?0 ?
19
Triple- Layer Model
-
?d
-
-
?d
-
??
Distance ?
??
OH O - OH OH2 OM L OH
-
?0
?0 ?
PowerShow.com is a leading presentation sharing website. It has millions of presentations already uploaded and available with 1,000s more being uploaded by its users every day. Whatever your area of interest, here you’ll be able to find and view presentations you’ll love and possibly download. And, best of all, it is completely free and easy to use.
You might even have a presentation you’d like to share with others. If so, just upload it to PowerShow.com. We’ll convert it to an HTML5 slideshow that includes all the media types you’ve already added: audio, video, music, pictures, animations and transition effects. Then you can share it with your target audience as well as PowerShow.com’s millions of monthly visitors. And, again, it’s all free.
About the Developers
PowerShow.com is brought to you by CrystalGraphics, the award-winning developer and market-leading publisher of rich-media enhancement products for presentations. Our product offerings include millions of PowerPoint templates, diagrams, animated 3D characters and more.