Applications of single fluorescent nanodiamonds FND as cellular biomarkers - PowerPoint PPT Presentation
1 / 13
Title:
Applications of single fluorescent nanodiamonds FND as cellular biomarkers
Description:
However, in experiment cell auto-fluorescence background is always a problem. ... a single FND particle bound with a single T4 DNA molecule on a glass substrate. ... – PowerPoint PPT presentation
Number of Views:212
Avg rating:3.0/5.0
Title: Applications of single fluorescent nanodiamonds FND as cellular biomarkers
1
Applications of single fluorescent nanodiamonds
(FND) as cellular biomarkers
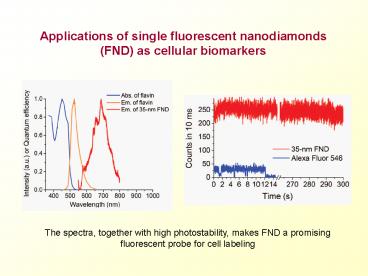
The spectra, together with high photostability,
makes FND a promising fluorescent probe for cell
labeling
2
- One of the key approaches to understand how
biological systems function is to probe
biomolecules individually and observe how they
interact with each other directly in vivo at
single molecule level. Fluorescent microscopy is
one of the techniques widely adopted to fulfill
this purpose, owing to its ultrahigh sensitivity.
However, in experiment cell auto-fluorescence
background is always a problem. Among all the
fluorophors used to label biomolecules such as
organic dyes and quantum dots, fluorescent
nanodiamond (FND) has drawn increasing attentions
primarily for the following reasons - As shown in left figure, the absorption and
emission spectrum of FND can well separate from
endogenous fluorescence emitted by intracellular
components thus with significantly reduced
background noise. - As shown in right figure, the tremendous
photostability with no photobleaching and no
blinking behavior makes it possible for long-term
observation.
3
Biomedical Applications of Single FND
Observation of a single FND particle bound with a
single DNA
Tracking of a single FND in a live cell
4
3. The surface of FND can easily be
functionalized with molecules such as nucleic
acids and proteins. As shown in left figure, a
single FND particle bound with a single T4 DNA
molecule on a glass substrate. 4. FND is
non-cytotoxic and bio-compatible. As shown in
right figure, a single FND can be tracked in a
live cell without killing cells. We have
testified all these qualities mentioned above in
our previous works using 35 nm FNDs. These
results were published on PNAS vol.104 p.727 and
were selected in the News of Analytical
Chemistry. We believe that this new material
shows highly potential in the filed of biomedical
and biotechnology.
5
Photoisomerization in two-dimension
- Bistilbene Photoisomerizationon a Surface
6
- We have observed stilbene photoisomerization on
the Ag/Ge(111)-v3 surface with STM. The direct
observation allows us to confirm the microscopic
one-bond-flip mechanism in the photoisomerization
reaction. A biexciton-assisted photoisomerization
model is proposed to explain the finding that the
surface photoisomerization reaction occurs in
pairs. The unique photochemistry observed here
not only is of intrinsic interest but also may
prove to be useful in understanding the workings
of singlemolecule optically activated
nanodevices. Also, the work was reported as J.
AM. CHEM. SOC. 2005, 127, 10788-10789.
7
Tomography of Laser-Wakefield Accelerators
programmed interaction length
variable-position knife-edge
pump pulse
electron energy spectrum
cylindrical lens pair
gas jet
machining pulse
setup for tomographic measurements
By using laser-machining to scan the interaction
length, we developed a tomographic measurement
technique for studying laser-plasma interaction
and applied it for the first time on
laser-wakefield accelerators.
8
Tomography of Laser-Wakefield Accelerators
uniform density region
up slope
down slope
electron central energy (MeV)
saturation
acceleration
position (?m)
In the laser-wakefield electron accelerator, from
the injection point (at 500 ?m) to the saturation
point (at 700 ?m) the energy of the electrons
increases to 45 MeV within a distance of 200 ?m.
9
Tomography of Laser-Wakefield Accelerators
We developed a tomographic measurement
technique for studying laser-plasma interaction
and applied it for the first time on
laser-wakefield accelerators. Our experiments
clarified the self-injection and acceleration
processes of electrons that produce
mono-energetic electron beams and gave the first
direct measurement of the 2.25-GeV/cm
acceleration gradient. We proved that after the
electron energy reaches saturation no
deceleration occurs. We also found that the
electron energy spectrum does not change after
self-injection. These observations are important
to the understanding of the working principles of
laser-wakefield accelerators. Papers are
published in Physical Review Letters 96, 095001
(2006), Physical Review E 75, 036402 (2007).
10
Are all forms of energy equal in promoting a
chemical reaction?
Impact of reactant C-H stretching excitation on
product images
11
The influence of vibrational excitation on
chemical reaction dynamics is well understood in
triatomic reactions, but the multiple modes in
larger systems complicate efforts toward a
predictive framework. By precise tuning
of translational energies, the team led by Kopin
Liu at IAMS recently measured the relative
efficiencies of vibration and translation in
promoting the gas phase reaction of CHD3 with Cl
atom to form HCl and CD3. Surprisingly, they
observed that C-H stretch excitation is no more
effective than an equivalent amount of
translational energy in raising the overall
reaction efficiency CD3 bend excitation is only
slightly more effective. Vibrational excitation
does have a strong impact on product state and
angular distributions, however, with C-H
stretch-excited reactants leading to
predominantly forward-scattered, vibrationally
excited HCl (IR-on Figure). This work is
published in Science 316, 1723-1726 (2007) a
perspective in the same issue (pp1707-1708)
commented on the significance and impact of this
work.
12
Nanoparticle enhanced optical spectroscopy
Left artists view of a nanoparticle-array with
many hot-junctions in between neighboring
particles, which are responsible for its
extremely high SERS enhancing power. Center SEM
image and TEM image (inset) of a Ag-nanoparticle
array on an AAO substrate. Right Integrated
Raman intensity of adenine at 739 cm-1 as a
function of interparticle gap width (W) for
different Ag/AAO substrates. Inset shows a
typical SERS spectrum of adenine.
13
When a molecule is irradiated by a green
light, it could emit very weak yellow light. This
is because a very small fraction of the green
light induces the vibration of the molecule and
loses part of its energy, or equivalently, the
frequency of the green light is decreased and its
wavelength is increased to become a yellow light.
Such shift in the frequency of the scattered
light is characteristic of the vibration
frequency of a molecule, similar to the
uniqueness of ones fingerprint. The identity of
a molecule can be identified by this so-called
Raman Spectroscopy. Prof. C. V. Raman, an
Indian scientist, was awarded the Nobel Prize in
Physics in 1930 for the discovery. Sponsored
by the National Science and Technology Program
for Nanoscience and Nanotechnology, a team of
researchers from the Institute of Atomic and
Molecular Sciences, Academia Sinica, National
Taiwan University, and National Yang-Ming
University, fabricated very ordered and
close-packed Ag nanoparticle arrays using
nanochannels on anodic aluminum oxide as the
template. When such a unique nanoparticle-array
is exposed to a laser, the field intensity on the
surface of and in the gaps between nanoparticles
is enhanced tremendously. For molecules adsorbed
on the array, their Raman signal intensity could
be enhanced by millions or even hundreds of
million times, therefore their chances of being
detected by Raman spectroscopy are greatly
improved. This technique based on
nanoparticle-array-enhanced Raman spectroscopy is
being exploited to detect various
environmentally, biologically, and bio-medically
important molecules. (Advanced Materials 18,
491-495 (2006))