Nitriles PowerPoint PPT Presentations
All Time
Recommended
20.18. Preparation of Nitriles. nucleophilic substitution by cyanide on. alkyl halides (Sections 8.1 and 8.13) cyanohydrin formation (Section 17.7) ...
| PowerPoint PPT presentation | free to download
Many carboxylic acids are found in Nature. Acetic Acid, CH3CO2H for vinegar ... Formic acid. Acetic acid. Propanoic acid. Propenoic acid. Benzoic acid ...
| PowerPoint PPT presentation | free to view
Starting materials for acyl derivatives (esters, amides, and acid chlorides) ... 20.2 Structure and Physical Properties of Carboxylic Acids ...
| PowerPoint PPT presentation | free to view
Nitriles are organic compounds having a triple bond between carbon and nitrogen. They are also known as cyanides. The most commonly occurring nitrile is Nitrile Rubber which is a synthetic copolymer of unsaturated acrylonitrile and conjugated butadiene. Such a form of rubber is highly resistant to chemicals
| PowerPoint PPT presentation | free to download
Electronegative substituents promote formation of the carboxylate ion. 11 ... Multiple electronegative substituents have synergistic effects on acidity. 12 ...
| PowerPoint PPT presentation | free to view
Carboxylic Acids and Their Derivatives Nucleophilic Acyl Substitution Carboxylic Acid Derivatives: Hydrolysis of an imine Reactions of Nitriles Addition of ...
| PowerPoint PPT presentation | free to view
reactions of organic compounds polymers dibromoalkanes ketones alkanes alkenes alcohols aldehydes esters halogenoalkanes amines carboxylic acids nitriles
| PowerPoint PPT presentation | free to download
a substituted acetoacetic ester. a substituted acetone ... Direct alkylation of ketones, esters, and nitriles (but NOT aldehydes) ...
| PowerPoint PPT presentation | free to view
Hydrolysis of Acid derivatives and Nitriles. Haloform reaction ... Thermolysis of beta-diacids. Thermolysis of beta-keto acids. Spectroscopy. Infrared. Pmr ...
| PowerPoint PPT presentation | free to view
Y = a leaving group -Cl, -O2CR', -OR', -OH, -NR2, 20.3: General Mechanism for Nucleophilic Acyl Substitution ... Cephalexin. 20.15: Preparation of Nitriles ...
| PowerPoint PPT presentation | free to download
Alkylation of Enolate Ions The malonic ester synthesis The acetoacetic ester synthesis Direct alkylation of ketones, esters and nitriles Relative acidity of selected ...
| PowerPoint PPT presentation | free to view
Amines via Reduction. Reduction of nitro compounds. Reduction of nitriles. Reduction of amides ... Amines via Bimolecular Nucleophilic Substitution. Amine Reactions ...
| PowerPoint PPT presentation | free to view
Dehydration synthesis of benzoic acid cyclopropylamine. Drill: ... Nitrile Water. R-C-N: H2O. Common. Nitriles. R-C=N. Acetonitrile. CH3-C=N. Hydrogen Cyanide ...
| PowerPoint PPT presentation | free to download
Carboxylic acids AH Chemistry, Unit 3 ... nitriles Reactions Neutralisation with bases and metals Producing salts Condensation with alcohols Producing esters Reaction ...
| PowerPoint PPT presentation | free to download
Mass of parent ion is molecular mass, not molecular weight. ... Aliphatic alcohols, amines, nitrites, nitrate, nitro compounds, nitriles, highly ...
| PowerPoint PPT presentation | free to view
Hydrolysis of Nitriles. The reaction can be conducted in either acidic or basic solution. ... Base-Promoted Hydrolysis of a Nitrile (Part One) WWU-Chemistry ...
| PowerPoint PPT presentation | free to view
N alcohols, aldehydes, ketones, nitriles, amides (5 carbons or less) ... pH(aqueous solution): _ . Department of Chemistry. University of Missouri-St. Louis ...
| PowerPoint PPT presentation | free to download
The increasing demand for sodium cyanide from the chemical industry represents one of the primary factors driving the global market. This compound is used in the derivation of several chemicals, including cyanogen chloride, cyanuric chloride, various nitriles, etc. Read more: https://www.syndicatedanalytics.com/sodium-cyanide-production-cost-analysis-report
| PowerPoint PPT presentation | free to download
Prsentation PowerPoint
| PowerPoint PPT presentation | free to view
CHE 242 Unit VIII The Structure, Properties, Reactions and Mechanisms of Carboxylic Acids and Their Derivatives CHAPTER TWENTY-ONE Terrence P. Sherlock
| PowerPoint PPT presentation | free to download
Use acetic formic anhydride to produce formate esters and formamides. ... Lactam Formation. Five- and six-membered rings can be formed by heating - and -amino acids. ...
| PowerPoint PPT presentation | free to view
Hydrolysis requires strong acid or base and heating under reflux. . It's not easy ! ... Base-Promoted Hydrolysis of a Amide. MECHANISM. stronger base. than OH ...
| PowerPoint PPT presentation | free to view
RCCl. O. least stabilized C=O. most stabilized C=O. lone ... RCCl. O. least stabilized C=O. most stabilized C=O. Sulfur (like chlorine) is a third-row element. ...
| PowerPoint PPT presentation | free to view
Nucleophilic ring opening of epoxides by ammonia (Section 16.12) ... As octylamine is formed, it competes with ammonia for the remaining 1-bromooctane. ...
| PowerPoint PPT presentation | free to download
name the acyl group and add the word chloride, fluoride, ... RCCl. O. RCOR' O. RCNR'2. O. Stabilization. very small. small. large. moderate. Relative rate ...
| PowerPoint PPT presentation | free to view
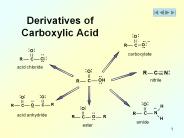
Derivatives of Carboxylic Acid carboxylate acid chloride nitrile acid anhydride amide ester Nomenclature of Acid Halides IUPAC: alkanoic acid alkanoyl halide ...
| PowerPoint PPT presentation | free to download
2. Nitrile compounds find extensive use in manufacturing of polymers and other chemicals. ... based on their substrate specificity as aliphatic or aromatic nitrilases. ...
| PowerPoint PPT presentation | free to view
the leading Nitrile Gloves Manufacturer look after the durability of the gloves. These gloves are three times more durable than latex gloves. They are FDA approved for medical use and higher puncture-resistant provide better protection to any contamination. These gloves are powder-free, so you need not worry about messy powder when working with adhesive or ink.
| PowerPoint PPT presentation | free to download
Acid + Ammonium ion Basic H2O and ... Reaction is driven to completion by the acid-base reaction ... Derivatives Characteristic Reactions Water and Acid ...
| PowerPoint PPT presentation | free to download
Cyanide addition. Section 16.7. Addition of Organometallics. CHEMISTRY OF ... Addition of Cyanide. Buffered to pH 6-8. In acid solution there would be little CN ...
| PowerPoint PPT presentation | free to view
Nomenclature. IUPAC: COOH group takes precedence ... Nomenclature. Structure & Properties. Synthesis. Reactions. Structure & Properties. Structure ...
| PowerPoint PPT presentation | free to view
Toxic (poisonous): Compounds that destroy life. or impair health by ... Glycosides of aliphatic nitrocompounds. Methylazoxymethanol (MAM) glycosides. Naringin ...
| PowerPoint PPT presentation | free to view
acetyl chloride. 3-methyl-4-pentenyl bromide. Chapter 21. 6. 7. Anhydrides - for symmetrical ... acetyl propionyl anhydride. Chapter 21. 7. B. Spectroscopy. 1. ...
| PowerPoint PPT presentation | free to view
Ethanoic methanoic anhydride ... if a 5- or 6-membered ring is the product. ethanoic anhydride acetic anhydride 1,2-benzenedicarboxylic anhydride phthalic anhydride ...
| PowerPoint PPT presentation | free to view
Aldol Condensation of Butanal. dehydration of -hydroxy aldehyde can be ... Electrophilic Alkenes. Termite Self-Defense! 18.12. Conjugate Addition to ...
| PowerPoint PPT presentation | free to view
Review of carbon atoms.
| PowerPoint PPT presentation | free to download
DROP -e FROM PARENT NAME AND ADD -ONE FOR A KETONE, -AL FOR AN ALDEHYDE ... THE MORE ELECTROPOSITIVE THE METAL, THE MORE IONIC THE CARBON - METAL BOND AND ...
| PowerPoint PPT presentation | free to view
treatment of a formic ester with 2 moles of Grignard reagent followed by ... 2. collapse of the TCAI gives a ketone (an aldehyde from a formic ester) 18. 18-52 ...
| PowerPoint PPT presentation | free to view
FOR DIACIDS, LONGEST CHAIN MUST INCLUDE BOTH -COOH GROUPS - KEEP -e AND ADD DIOIC ACID ... BUTYRIC ACID. BENZOIC ACID, PHTHALIC ACID. MALONIC ACID, ACETOACETIC ACID ...
| PowerPoint PPT presentation | free to view
... molecular-weight acid anhydrides react readily with water to give two ... most commonly used are sodium or potassium salts. 18-47. Interconversions. 18-48 ...
| PowerPoint PPT presentation | free to view
Title: Project Overview Author: USER Last modified by: bryson Created Date: 11/7/2001 3:36:02 PM Document presentation format: On-screen Show Company
| PowerPoint PPT presentation | free to download
19.2: Structure and Bonding (please read) 19.3: Physical Properties. ... contains both a hydrogen bond donor (-OH) and a hydrogen. bond acceptor (C=O) ...
| PowerPoint PPT presentation | free to download
The carboxyl group (-CO2H) is the parent group of a family of compounds called ... In IUPAC nomenclature, the name of a carboxylic acid is obtained by changing the ...
| PowerPoint PPT presentation | free to download
32.2 Nomenclature of Carboxylic Acids and their Derivatives ... Boiling the esters under reflux with dilute HCl or dilute H2SO4 give the parent ...
| PowerPoint PPT presentation | free to view
Ester ... acid chloride, acid anhydride, ester ?????????????????????? aldehyde ???? ketone ... ester C2-C5, amide C1-C6 ???????????. ??????????????????????????? ...
| PowerPoint PPT presentation | free to download
Title: PowerPoint Presentation Author: mucin Last modified by: Hudecz Ferenc Created Date: 1/26/2004 9:52:56 AM Document presentation format: On-screen Show
| PowerPoint PPT presentation | free to download
Click a box below to go to the mechanism Click here for advice AS Free Radical Substitution Electrophilic Addition Nucleophilic Substitution (nitrile hydrolysis reaction)
| PowerPoint PPT presentation | free to view
... A cyclic ether containing two carbons is called ethylene oxide ... beverage is merely twice the ... Physical Properties of Aldehydes and Ketones Carbon-oxygen ...
| PowerPoint PPT presentation | free to download
Physical Properties. Melting Point. Boiling Point. Density. Solubility. Refractive Index ... wavelength of radiation absorbed are properties of a group of atoms ...
| PowerPoint PPT presentation | free to view
Use Conjugated Molecules; Carbonyl Group, Nitro Group ... The compound does not contain 'Carbonyl' or 'Nitro' groups. ... Double Bonds, Triple Bonds and Nitro Groups ...
| PowerPoint PPT presentation | free to view
Global performance elastomers market size is expected to reach $25.62 Bn by 2028 at a rate of 10.7% segmented as by type, nitrile-based elastomers, fluoroelastomers
| PowerPoint PPT presentation | free to download
Conquest Distributors provides high-quality Nitrile Gloves for reliable protection. Our gloves offer superior durability, comfort, and flexibility for various applications. From leather work to disposable options, find the best prices on Nitrile, Disposable, and vinyl gloves. Order now for the best selection of nitrile gloves to keep yourself and others safe. Contact us at (416) 848-0092 for more information today!
| PowerPoint PPT presentation | free to download
Made from premium materials: Choose from nitrile, latex, or polyethylene cots, each with its own unique properties to meet your specific needs.
| PowerPoint PPT presentation | free to download
Superior Nitrile Gloves – Powder Free have been developed from a low particulate synthetic nitrile material that enables them to be strong and durable while remaining light and extremely comfortable. The very low ionic extractable levels and particulate generation present in these gloves make them an ideal choice for critical environments.
| PowerPoint PPT presentation | free to download
Experience the difference with our nitrile gloves that are manufactured under the stringent European quality control standards by a leading Nitrile Glove Manufacturer. Our partnership with a top US Medical Glove Company ensures we deliver nothing but the best nitrile gloves to you. An eco-conscious choice, our glove is a biodegradable Nitrile Glove, emphasizing our commitment towards a sustainable future. As one of the top gloves manufacturers, we pride ourselves on providing the finest quality nitrile gloves to all our customers.
| PowerPoint PPT presentation | free to download
Nitrile Gloves Market
| PowerPoint PPT presentation | free to download