Training%20plain%20background PowerPoint PPT Presentation
Title: Training%20plain%20background
1
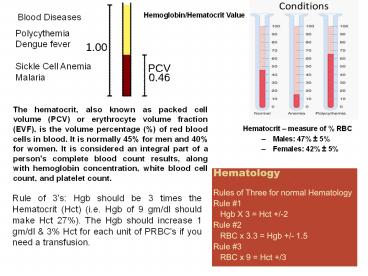
Hemoglobin/Hematocrit Value
Blood Diseases
Polycythemia Dengue fever Sickle Cell Anemia
Malaria
The hematocrit, also known as packed cell volume
(PCV) or erythrocyte volume fraction (EVF), is
the volume percentage () of red blood cells in
blood. It is normally 45 for men and 40 for
women. It is considered an integral part of a
person's complete blood count results, along with
hemoglobin concentration, white blood cell count,
and platelet count.
- Hematocrit measure of RBC
- Males 47 5
- Females 42 5
Rule of 3's Hgb should be 3 times the Hematocrit
(Hct) (i.e. Hgb of 9 gm/dl should make Hct 27).
The Hgb should increase 1 gm/dl 3 Hct for each
unit of PRBC's if you need a transfusion.
2
Introduction To Rheology
Santanu Dhara School of Medical Science and
Technology Indian Institute of Technology Kharagpu
r-721302
3
Definition of Rheology
- Rheology is the science of flow and deformation
of matter - We use rheology to study flow behavior of
ceramics loaded slurries for preparation, casting
and gelling.
4
Flow and Deformation Parameters Shear Stress,
Shear Strain, Shear Rate
5
Range of Rheological Material Behavior
- Rheology The study of flow and deformation of
matter. - Range of material behavior
- Solid Like ---------Liquid Like
- Ideal Solid-------------Ideal Fluid
- Classical Extremes
6
Classical Extremes Elasticity
- 1678 Robert Hooke develops his
- True Theory of Elasticity
- The power of any spring is in the same
proportion with the tension therof. - Hookes Law ? G? or (stress G x strain)
- where G is the RIGIDITY MODULUS
- Hookes law describes ideal mechanical behavior
using a constitutive equation in which stress and
strain are related through a proportionality
constant called the modulus G. If you double the
stress, you double the strain.
7
Classical Extremes Viscosity
- 1687 Isaac Newton addresses liquids and steady
simple shearing flow in his Principia - The resistance which arises from the lack of
slipperiness of the parts of the liquid, other
things being equal, is proportional to the
velocity with which the parts of the liquid are
separated from one another. - Newtons Law ? ??
- where???is the Coefficient of Viscosity
- Newtonss law describes idea flow behavior using
a constitutive equation in which stress and rate
of strain are related through a proportionality
constant called the viscosity. If you double the
stress, you double the shear rate.
8
Viscosity and Steady Shear Testing
9
Viscosity Definition
- Viscosity is . . . .
- lack of slipperiness.
- synonymous with internal friction.
- resistance to flow.
A measure of the resistance of flow due to
internal friction when one layer of fluid is
caused to move in relationship to another layer.
10
Viscosity Units
- The Units of Viscosity are . . . . .
- SI unit is the Pascal.second (Pa.s)
- cgs unit is the Poise
- Poise is gt Pa.s by a factor of 10
- 10 Poise 1 Pa.s
- 1 cP (centipoise) 1 m Pa.s (mili-pascal-second)
11
- Typical Viscosities (Pa.s)
- Asphalt Binder ---------------
- Polymer Melt -----------------
- Molasses ----------------------
- Liquid Honey -----------------
- Glycerol -----------------------
- Olive Oil -----------------------
- Water --------------------------
- Air -------------------------------
100,000 1,000 100 10 1 0.01 0.001 0.00001
12
- Typical Shear Rates (1/s)
- Sedimentation 10-4
- Molecular Structure
- Leveling/Sagging 10-3 to 100
- Compression Molding
- Pouring 100 to 101
- Extrusion
- Pumping 101 to 103
- Blow Molding
- Rubbing 103 to 104
- Injection Molding
- Spraying 105
- Bearing lubrication 106
13
Variables that Affect Viscosity
- Shear Rate
- Time of Shearing
- Temperature
- Pressure
14
Newtonian Vs. Non-Newtonian Behavior
- Strict definition of Newtonian Behavior
- ? is only stress generated in simple shear flow
(no normal stress difference). - Shear viscosity does not vary with shear rate.
- ? is constant with time of shearing.
- ? in fluid falls immediately to zero when
shearing is stopped. When sheared again, the
??is as was previously measured (regardless of
delay between measurements). - ? measured in different types of deformation are
in proportion to one another. - ??measured in uniaxial extension is three times
shear ? (Troutons Ratio) - A Liquid showing any deviation from Newtonian is
said to be non-Newtonian
15
Characteristic Diagrams for Newtonian Fluids
?, Pa
???Pa.s
16
Non-Newtonian Fluids
- Non-Newtonian Time Independent Liquids, ? ?(??)
- Viscosity of fluid is dependent on shear rate but
independent of the time of shearing. The
viscosity is presented at a specific shear rate
and referred to as the apparent viscosity,
shear viscosity or shear dependent viscosity. - Non-Newtonian Time Dependent Liquids, ? ?(??t)
- Viscosity of fluid is dependent on shear rate and
the time during which shear rate is applied.
17
Non-Newtonian, Time Independent Fluids
- Shear-Thinning
- A decrease in viscosity with increasing shear
rate. Also referred to as Pseudoplasticity. - Shear-Thickening
- An increase in viscosity with increasing shear
rate. Also referred to as Dilatancy.
18
Characteristic Diagrams for Shear Thinning Fluids
19
Shear Thinning Behavior
- Shear thinning behavior is often a result of
- Orientation of non-sherical particles in the
direction of flow. An example of this phenomenon
is the pumping of fiber slurries. - Orientation of polymer chains in the direction of
flow and breaking of polymer chains during flow.
An example is polymer melt extrusion - Deformation of spherical droplets to elliptical
droplets in an emulsion. An industrial
application where this phenomenon can occur is in
the production of low fat margarine. - Breaking of particle aggregates in suspensions.
An example would be stiring paint.
20
Non-Newtonian, Time Dependent Fluids
- Thixotropy
- A decrease in apparent viscosity with time under
constant shear rate or shear stress, followed by
a gradual recovery, when the stress or shear rate
is removed. - Rheopexy
- An increase in apparent viscosity with time under
constant shear rate or shear stress, followed by
a gradual recovery when the stress or shear rate
is removed. Also called Anti-thixotropy or
negative thixotropy.
ReferenceBarnes, H.A., Hutton, J.F., and
Walters, K., An Introduction to Rheology,
Elsevier Science B.V., 1989. ISBN 0-444-87469-0
21
Non-Newtonian, Time Dependent Fluids
Shear Rate Constant
Rheopectic
Viscosity
Thixotropic
time
22
Linearity vs. Non-Linearity
- Hookes and Newtons laws are linear laws. They
assume direct proportionality between stress and
strain, or shear rate no matter what the stress. - Most materials we work with obey these laws over
a limited range of stresses. Beyond this limited
range a material behaves non-linearly.
23
Newtonian and Non-Newtonian Behavior of Fluids
Non-Newtonian Region ? f(?)
?
s
?
s
24
Linear and Non-Linear Stress-Strain Behavior of
Solids
1000
100.0
Non-Linear Region G f(?)
Linear Region G is constant
100.0
G
G' (Pa)
osc. stress (Pa)
10.00
s
0.01000
1.000
1000.0
0.010000
0.10000
1.0000
10.000
100.00
strain
25
- Types Of Flow
"Yield"
Bingham Plastic Pseudoplastic Newtonian Dilat
ant
s
Shear Thinning
Yield Stress
Shear Thickening
26
- Model Fitting
Newtonian Pseudoplastic Dilatant Bingham Casso
n Herschel-Bulkley
27
Steady Shear Test Modes
- Stepped Ramp - Equilibrium Flow
- Continuous Ramp
- Temperature Ramp
28
- Stress Ramp Test - Continuous Ramp
- Stress is applied to material at a constant
rate. Resultant strain is monitored with time.
- USES
- Yield stress
- Scouting Viscosity Run
29
- Idealized Full Flow Curve
(1)
(1) Sedimentation (2) Leveling (3) Pouring (4)
Pumping (5) Rubbing (6) Spraying
Asphalt Binder
(2)
Molasses
(3)
Glycerol
Log h
Castor Oil
(4)
(5)
(6)
Olive Oil
Water
10 E 4
10 E - 6
10 E 1
.
Log g
30
- Viscosity Temperature dependence
31
Type of Viscometer
- Ostwald viscometers named after Wilhelm Ostwald
or glass capillary viscometers. Another type is
the Ubbelohde viscometer. - They basically consist of a glass tube in the
shape of a U held vertically in a controlled
temperature bath. - In one arm of the U is a vertical section of
precise narrow bore (the capillary). Above this
is a bulb, there is another bulb lower down in
the other arm. - In use, liquid is drawn into the upper bulb by
suction, then allowed to flow down through the
capillary into the lower bulb. - Two marks (one above and one below the upper
bulb) indicate a known volume. The time taken for
the level of the liquid to pass between these
marks is proportional to the kinematic viscosity.
- Most commercial units are provided with a
conversion factor, or can be calibrated by a
fluid of known properties
32
- Rotary Viscometer - Spring
- Brookfield
- Fungilab viscoelite
- Air bearing (Graphite bearing)
- Bohlin Rheometers and Viscometers
- TA instrument
33
- CSL2
Air Bearing Motor Optical Encoder Measuring
System Temperature Control Cell Autogap Set
2
CSL
100
Rheometer
34
- Cross-Section CSL
Draw Rod
Optical Encoder
Air Bearing
Controlled Torque Motor
Measurement Geometry
Peltier Temperature Control Plate
Micrometer Wheel
Pneumatic Ram
Auto Gap Set Motor
35
- Cross-Section of CSL Drive
Air Bearing
Optical Encoder
Air Jet
Air Jet
Non-Contact Motor
Drag Cup
36
- Controlled Stress Schematic
37
- AR 1000 Rheometer Head Schematic
Optical Encoder
Thrust Bearing
Drag-cup Motor
Motor Housing
38
Parallel Plate
- Variable Gap (Sample Thickness)
- 500 to 2000 microns recommended.
- Assortment of Plate Diameters (2 cm, 4 cm, and 6
cm Standard) - Easy to load. Able to use with a wide range of
viscosities. - Velocity gradient from center to edge of plate
during steady shear testing.
39
- Plate Gaps and Diameters
Shear Stress
2cm
4cm
6cm
Decreases
Gap Shear Rate
0 Infinity
Increases
40
Gap Choice for Parallel Plate Geometry
- Set gap to be at least 10 x particle or droplet
- size consider extremes of size distribution
- Minimum gap should be 1000 microns
41
- Cone Angles and Diameters
Shear Stress
2cm
4cm
6cm
Decreases
Angle Shear Rate
Increases
42
- Limitations of Cone Plate for Dispersions -
Fixed Gap!
Truncation Heights 1 degree 20 - 30 microns 2
degrees 60 microns 4 degrees 120 microns
Truncation Height Gap
Cone Plate
Gap must be gt or 10 particle size!!
43
Concentric Cylinder
- Large surface area to obtain low stress
measurements. - Possibility of shear history effects from
loading. - Good for testing suspensions with limited
stability.
44
Solvent Trap System
- Reduces errors due to solvent evaporation
- Available for cones, plates, and concentric
cylinders
45
- Edge Effects
Under Filling
Over Filling
Correct Filling
46
Rheology of Dispersions
47
Dispersion - Definition
- DEFINITION Discrete particles randomly
distributed in a fluid medium. Dispersions can
be broken into three categories - Suspensions - solid particles in a liquid medium.
- Emulsions - liquid droplets in a liquid medium.
- Foam - a gas in a liquid medium.
ReferenceMacosko, C.W., Rheology Principles,
Measurements, and Applications, VCH Publishers
Inc., 1994. ISBN 1-56081-579-5
48
Dispersion
Particle, Droplet, or Air Bubble
Number, Size, Shape, Density
Modify Surface
-
Liquid Medium
Continuous Phase, hc
49
Factors Influencing Rheology of Dispersions
50
Concentration of Particles
- Rheology depends greatly on the hydrodynamic
- forces that act on the surface of particles
(aggregates) regardless of the density. - Need to define concentration of suspension in
terms of phase volume or volume-per-volume
fraction ??as opposed to weight-per-weight
fraction which is often used as a measure of
concentration.
ReferenceBarnes, H.A., Hutton, J.F., and
Walters, K., An Introduction to Rheology,
Elsevier Science B.V., 1989. ISBN 0-444-87469-0
51
Forces Acting on Particles
- Arise from interaction between particles and
result in overall - repulsion or attraction between particles.
- Repulsion
- Like electrostatic charges.
- Entropic repulsion of polymeric or surfactant
material on the - surface of the particle.
- Net repulsive Forces ??Particles remain separate
(dispersed or deflocculated). - Attraction
- London - Van der Walls attraction between
particles. - Electrostatic attraction between unlike charges
on different parts of a particle (edge/face
attraction between clay particles). - Net attractive forces ??Particles flocculate.
ReferenceBarnes, H.A., Hutton, J.F., and
Walters, K., An Introduction to Rheology,
Elsevier Science B.V., 1989. ISBN 0-444-87469-0
52
- Forces Between Particles
- In most dispersions, particles are kept apart (in
suspension) by modifying the surface of the
particle.
Van der Waals Forces lead to particle clumping
Want to prevent particles from clumping
- There are generally two ways to modify the the
surface of a particle to maintain a stable
dispersion - Electrostatics forces
- Steric forces
53
Mechanism of stabilization of the colloidal
particle in suspending medium
UT UA (URe URs ) Where URe and URs are
the energy term due to electrostatic repulsion
and steric stabilization
The repulsive force should be predominant over
Vander Walls force of attraction
For electro statically stabilized slurry, the
zeta potential of the suspended powder should be
higher than 25 mV
54
Different State of Dispersion of Slurry
- Flocculated Slurry
- Dispersed Slurry
- Coagulated Slurry
55
Slurry Characteristics
- Forming of dense, defect free ceramic components
via gelcasting requires use of highly loaded
slurries. - In addition to high solids loading, gelcasting
slurries must be stable, Shear thinning - Yield stress ? 50 Pa
- Viscosity ? 2 Pa.s at the shear rate 10 s-1
56
Dispersion Stability
- Two ways dispersed particles are stabilized.
- Electrostatically
- The existence of a net charge which causes
particles to repel one another. - Sterically by absorption of polymer molecules on
the particles. - film of absorbed surfactant which which prevents
the particles from adhering to one another may be
sufficient to keep dispersed particles in
suspension.
Ref Rohn, C.L.,The Rheology of Coatings and
Dispersions, Journal of Water Borne Coatings, Au
gust, 1987.
57
Electrostatic Forces on Particles
Electrostatic Repulsion
Pseudo-Structure
58
- Steric Hindrance of Particles
Adsorbed Polymer
Non-interacting
Bridging Floc
59
Concentrated Suspensions
- In concentrated suspensions, particles may link
to build a three-dimensional network structure
which extends through the whole system. - This structure in a suspension is a result of
particle-particle interactions. These
particle-particle interactions cause deviation
from Newtonian behavior in a suspension. - We need to recognize that the rheology of
suspensions is measured macroscopically but the
rheology depends very much on microscopic
considerations. Measuring these
particle-particle interactions or structure will
yield valuable information about the
microstructure of the suspension.
Reference Sato, T., Rheology of Suspensions,
Journal of Coatings Technology, Vol. 67, No.
847, August 1995.
60
Particle-Particle Interactions Cause Structure
particle-particle interactions
61
The Maximum Packing Fraction, ?m
- The maximum packing fraction ?m is the volume
fraction of particles at which a
three-dimensional continuous contacting network
is formed. - At ?m the suspension is jammed up and flow is
impossible. - At ?m the suspension the viscosity goes to
infinity. - ?m ranges form 0.5 to 0.75. (FCC ?m 0.74)
62
The Maximum Packing Fraction, ?m
Packing Fraction The ratio of the total volume
of a set of objects packed into a space to the
volume of that space.
- ?m depends on particle arrangement, particle size
distribution, and particle shape. - Broader particle size distributions tend to have
higher ?m. the suspension is jammed up and flow
is impossible. - Non-spherical particles have lower ?m (poor
space fitting). - Particle flocculation leads to a low ?m (in
general flocs are not close packed). - Good to normalize concentration as the
ration??/?m.
63
Limited Cases for the Viscosity FlowCurve for
Suspensions
Yield stress develops below which there is no
real macroscopic flow
Low shear viscosity limit often disappears with
higher concentration
log ?
log t
64
Ink Flow Curves h vs. Shear Stress
Yield stress develops below which there is no
real macroscopic flow
1000000
100000
Low shear viscosity limit often disappears with
higher concentration
10000
viscosity (Pa.s)
1000
100.0
10.00
1.000
0.1000
1000
1.000
10.00
100.0
shear stress (Pa)
65
Ink Flow Curve h vs. Shear Stress
66
- "Yield" Curve for Grease
400 Pa
600 Pa
1.0E8
1.0E7
800 Pa
1.0E6
1.0E5
Six Decade Drop in h
viscosity (Pa.s)
1.0E4
1.0E3
1.0E2
10.00
1.0
10000
100.0
1000
shear stress (Pa)
67
The yield strength or yield point of a material
is defined in engineering and materials science
as the stress at which a material begins to
deform plastically. Prior to the yield point the
material will deform elastically and will return
to its original shape when the applied stress is
removed. Once the yield point is passed some
fraction of the deformation will be permanent and
non-reversible.
How do you measure?
68
Yield Stress of Alumina-Zirconia Suspensions
The yield stress of concentrated suspensions of
alumina, zirconia, and mixed alumina-zirconia
powders was measured by the vane technique as a
function of solids loading, relative amounts of
alumina and zirconia, and pH. At the isoelectric
point (IEP), the yield stress varied as the
fourth power of the solids loading.
Vijay Ramakrishnan, Pradip S. G. Malghan using
Vane geometry.
J. Am. Ceram. Soc, 1996.
69
Ink Flow Curves h vs. Shear Rate
t 10.5 Pa
t 11.5 Pa
70
Concentrated Suspensions Exhibit Elasticity
- Concentrated suspensions typically show some
degree of elasticity. If the material has a
yield stress then it behaves as an elastic gel
under small stresses or strains. - The storage modulus, G, is a measure of the
elasticity of the material and is a direct
measure of the particle-particle interactions.
Hence, G is a measure of the structural
characteristics of a material. - We can therefore use Dynamic Mechanical Testing
to characterize the structure of a concentrated
suspension. The mechanical response is extremely
sensitive to the amplitude of the applied
deformation, ? or ?.
71
Gel Structure in Suspensions
Suspension Gel Structure
72
Gel Structure in Suspensions
Structure begins to Breakdown
Unbroken Gel Structure
Broken Structure
Increasing Amplitude of Shear Deformation
73
Stability is Related to Structure in Inks
74
Time-Dependent Gellation
Gel Structure rebuilds with time
Break Structure under high shear
Time
75
Optimization of the dispersant
Darvan
DBAC
- The optimized dispersant amounts - 4.6 mg and 1.9
mg Darvan and DBAC respectively per gm of alumina
powder - 55 vol alumina loading slurry showed shear
thinning behaviour
76
Effect of nature and dispersant amount on
non-Newtonian index
The zeta potential values were maximum for
optimum amount of dispersant The zeta potential
value was high enough to stabilize the slurry
- All slurries were pseudoplastic in nature (n lt1)
- The non-Newtonian index n as a function of
dispersant amount
- All slurries had n values in the range of 0.11
to 0.33. - n was maximum for the optimum amount of
dispersant.
77
Comparison of the two dispersants
DBAC
Darvan
- For the same percent mole fraction below optimum,
the increase in viscosity was sharper for DBAC
slurries as compared to the Darvan slurries. This
was because the carboxylic group of Darvan is 15
times that of DBAC and the ability of Darvan to
provide steric hindrance. - Sharper rise in viscosity was observed for
similar percent mole fraction above the optimum
in case of Darvan in comparison to DBAC due to
tangling and double layer compression
78
Aging Behaviour of the slurries
DBAC
Darvan
- Among all Darvan and DBAC based slurries, the
ones with below optimum amount of dispersant
exhibited significant change in viscosity as a
result of both static and dynamic aging
treatments. - Slurries with optimum and above optimum amounts
of the two dispersants exhibited much lesser
change in viscosity as a result of static or
dynamic aging treatments.
J. Am. Ceram. Soc, 2005
79
Bacterial Aging
80
Hysteresis behaviour
- Hysteresis was maximum for less than optimum
amount of the dispersants and minimum for optimum
amount of the dispersants. - For more than optimum amount of dispersant,
hysteresis was observed for Darvan based slurries
due to tangling of long chain dispersant while
for DBAC based slurries no hysteresis was
observed.
81
Darvan
DBAC
Thixotropic behavior of aged slurries followed
similar trends as exhibited by the changes in
viscosity. Slurries that experienced an increase
in viscosity also showed pronounced thixotropic
behavior following aging.
82
Summary of the Aging of gelcasting slurries
- 55 vol alumina loading slurries were shear
thinning in behaviour - The slurries with optimum amount dispersants were
most stable due to maximum zeta potential values
thus viscosity was minimum. - At optimum level of dispersant the n values
were maximum. - The hysteresis behaviour was minimum with optimum
level of dispersants. - Hysteresis of aged slurries was similar to aging
behaviour of slurries
83
- Below optimum amount of dispersant exhibited
significant change in viscosity as a result of
both static and dynamic aging treatments. - During static aging treatments, the formation of
inter-particle network was responsible for the
observed increase in viscosity. - Roll mixing of slurries (dynamic aging) led to a
decrease in viscosity for Darvan based slurries,
similar change in viscosity was not observed for
DBAC based slurries. - Rather an increase in viscosity was observed for
DBAC based slurries, most likely due to
bio-degradation of DBAC during the dynamic aging
period at below optimum level of dispersant. - It was concluded that in general dispersant
addition above the optimum level may be tolerable
but care must be taken to not let the dispersant
addition be below the optimum.
84
Different terms related to Rheology
Shear Stress Shear strain or Strain
rate Viscosity Yield Stress Newtonian
Non-Newtonian Non-Newtonian Index Consistency
factor Pseudoplastic Plastic Bingham Power law,
Casson, Herchele Buckley and other different
models
Slippage Different geometry Solvent
trap Thixotropy Rheopexy Shear Thinning
(Pseudoplastic) Shear thickening
(Dilatant) Instantaneous viscosity Apparent
Viscosity Aging Structure of material
85
- Linear Viscoelasticity
86
- Viscoelasticity Defined
Range of Material Behavior Solid Like ----------
Liquid Like Ideal Solid ----- Most Materials
----- Ideal Fluid Purely Elastic -----
Viscoelastic ----- Purely Viscous
Viscoelasticity Having both viscous
and elastic properties
87
- Linear Viscoelasticity Defined
"If the deformation is small, or applied
sufficiently slowly, the molecular arrangements
are never far from equilibrium. The mechanical
response is then just a reflection of dynamic
processes at the molecular level which go on
constantly, even for a system at equilibrium.
this is the domain of LINEAR VISCOELASTICITY.
The magnitudes of stress and strain are related
linearly, and the behavior for any liquid is
completely described by a single function of
time." (Written by Bill Graessley, Princeton
University)
Reference Mark, J.,et.al., Physical Properties
of Polymers, American Chemical Society, 1984, p.
102.
88
- Response for Classical Extremes
Spring
Purely Viscous Response Newtonian Liquid ? ??
Dashpot
Purely Elastic Response Hookean Solid ?s G?
In the case of the classical extremes, all that
matters is the values of stress, strain, strain
rate. The response is independent of the
loading.
89
- Time-Dependent Viscoelastic Behavior
- Solid and Liquid Properties of "Silly Putty"
T is long 24 hours
T is short lt 1s
Deborah Number De ??/ ?
90
- Dynamic Mechanical Analysis
- OR
- Oscillatory Testing
91
- Dynamic Mechanical Testing
- An oscillatory (sinusoidal)
- deformation (stress or strain)
- is applied to a sample.
- The material response
- (strain or stress) is measured.
- The phase angle ?, or phase
- shift, between the deformation
- and response is measured.
92
- Dynamic Mechanical Testing
- Response for Classical Extremes
Purely Viscous Response (Newtonian Liquid)
Purely Elastic Response (Hookean Solid)
? 90
? 0
Stress
Stress
Strain
Strain
93
- Dynamic Mechanical Testing Viscoelastic Material
Response
Phase angle 0 lt d lt 90
Strain
Stress
94
Viscoelastic property of a slurry or gel
The viscoelastic property is characterized using
sinusoidal applied shear stress, ? ?o sin
?t ? shear stress, ?o, shear stress amplitude,
? angular velocity and t time.
95
The resulting deformation (?) developed in the
slurry can be represented by
? ?o sin (?t ?) ?o
strain amplitude and ? phase displacement angle
between applied stress and resulting deformation.
- 90o indicates purely viscous behaviour when
stress is out of phase with strain - 0o indicates a purely elastic behaviour when
stress and strain are in phase - 45o indicates a purely viscoelastic character
of the slurry
96
The complex modulus G is defined as G
?o / ?o
The complex modulus is divided into elastic
storage modulus (G') and viscous loss modulus
(G?) as follows G' G cos d G? G sin
d The storage modulus (G') is a measure of
energy stored elastically, while the loss modulus
(G?) is a measure of the energy dissipated as
heat during flow in the system after a shear
perturbation. The loss tangent (tan d) can be
obtained from the ratio of loss modulus and
storage modulus i.e., tan d G?/G'.
Most of the slurries are non-Newtonian and
exhibit both viscous flow and elastic behaviour,
which can be characterized by the above
measurements. The viscoelastic measurements can
also be used for studying gelation kinetics or
coagulation with change in ionic strength in the
slurries.
97
- DMA Viscoelastic Parameters
- The Complex, Elastic, Viscous Stress
- The stress in a dynamic experiment is referred
to as the complex stress ?
- The complex stress can be separated into two
components - 1) An elastic stress in phase with the strain.
?' ?cos?? - ??????' is the degree to which material behaves
like an elastic solid. - 2) A viscous stress in phase with the strain
rate. ?" ?sin? - ??????" is the degree to which material behaves
like an ideal liquid.
Phase angle d
? ?' i?"
Complex Stress, ?
Strain, ?
98
- DMA Viscoelastic Parameters
The Complex Modulus Measure of materials
overall resistance to deformation.
G Stress/Strain G G iG
The Elastic (Storage) Modulus Measure of
elasticity of material. The ability of the
material to store energy.
G' (stress/strain)cos?
The Viscous (loss) Modulus The ability of the
material to dissipate energy. Energy lost as
heat.
G" (stress/strain)sin?
Tan Delta Measure of material damping - such
as vibration or sound damping.
Tan ?? G"/G'
99
- DMA Viscoelastic Parameters Damping, tan ?
Dynamic measurement represented as a vector It
can be seen here that G (G2 G2)1/2
- The tangent of the phase angle is the ratio of
the loss modulus to the storage modulus.
tan ? G"/G'
- "TAN DELTA" (tan ?)?is a measure of the damping
ability of the material.
100
Gel point !!!
- Cross-over point of G' and G? is the gel point
over time and temperature.
101
- Application
- Viscoelastic behaviour of the slurry
- Gelation Kinetics
- Structure of the slurry
102
How to measure gelation kinetics?
- Oscillation behaviour of the sample is examined
to understand gelation kinetics by studying
different parameters such as elastic modulus
(G?), viscous modulus (G?) and phase angle (?) at
different conditions. - For carrying out measurement of gelation
kinetics, the following steps are performed- - The first step is to find out the constant values
of strain and frequency where the strain
(deformation) is proportional to the stress, and
G? and G? are independent of the measuring
conditions (stress, strain, and frequency). Thus
linear viscoelastic behaviour of the slurry
against time sweep measurement.
103
How to find out linear visco-elastic region?
- Amplitude sweep measurement of G G vs. strain
(0.001 to 100 according to material) at constant
frequency (1 Hz and constant temperature). Look
at the linear region in the plot of G G vs.
strain. Find out the value of strain in the 2/3
of the linear region. - G G vs. frequency sweep (1 to 100 Hz)
measurement at constant strain (selected from
2/3 of the linear region from above). Find out
the value of frequency in the 2/3 of the linear
region. - Now, measure G G vs. time sweep at constant
strain and frequency. This measurement will be a
linear plot as strain and frequency are within
the limit of elastic deformation zone. - Plot of G G vs. time or temperature soon
after addition of cross linking agent will be
resulted in change in phase deference delta and
increase in storage modulus and reduction in loss
modulus. At delta0 degree, gelation is complete.
104
First, amplitude sweeps were performed at 1 Hz on
0.5 wt chitosan solution. The amplitude sweeps
showed G? and G? to be strain independent at
deformations between 0.001 and 10. Based on this
result of amplitude sweep, a value of 1.5 strain
was chosen in the linear regime for frequency
sweep measurement. From frequency sweep
measurement at constant 1.5 strain, a value of 2
Hz was chosen in the linear regime. Time sweep
and temperature sweep measurement were carried
out to examine gelation behaviour of the 0.5 wt
aqueous chitosan solution at constant strain of
1.5 and 2 Hz frequency. Time sweep measurement
showed linear viscoelastic region at applied
strain and frequency with comparable magnitude of
G? and G? values. This result indicates that 0.5
wt aqueous chitosan has almost 50-50 solid
liquid behaviour. When the same experiment was
carried out with temperature sweep measurement
(Fig.3), it was observed that both elastic
modulus (G?) and viscous modulus (G) are going
up after 50o C temperature and phase angle also
started going down and finally become almost zero
degree. The magnitude of increase in G? was
relatively higher than G? due to temperature
assisted gelation of chitoan-DHF system. After
completion of the measurement, it was observed
that the chitosan sample becomes brown film due
to gelation reaction.
105
(No Transcript)
106
(No Transcript)
107
(No Transcript)
108
(No Transcript)
109
(No Transcript)
110
(No Transcript)
111
Gelcasting of polymerizable MAM-MBAM based slurry
- The grinding media was removed and the slurries
were well deaired - Prior to mold filling, initiator Ammonium per
sulphate (APS) and catalyst Tetra methyl ethylene
diamine (TEMED) were added and mixed - Slurries were poured into metal or plastic molds
coated with white petroleum jelly - The molds containing the slurries were kept in an
oven preheated to 50oC for gel formation.
- The parts were removed from the mold soon after
gelling before any significant drying could occur
112
Gelation behaviour of 55 volume alumina
slurrries
Temperature Induced Gelation
Room Temperature Gelation
Measurement conditions Frequency 1 Hz Strain 1
113
Environment friendly process for shaping of
ceramics
- Homogenize extracted egg white
- Prepare egg white-water premix, add antifoaming
agent (1-octanol) - Add dispersant, powder and mill for 24 hours
- Filter the slurry to separate the media, roll mix
for an hour and idle for 30 minutes - Cast in molds and place in oven or water bath
preheated to 80oC
Temperature induced gelation of a slurry with 30
volume ceramic and 50 binder in premix
S. Dhara and P. Bhargava, J. Am. Ceram. Soc., 84
12 3048-50 (2001)
At Frequency 1 Hz, Strain 1
114
Rheological Characteristics of Egg White based
Slurry
35 vol powder
- For same binder water ratio, viscosity of the
slurry increased with the increase in solids
loading - For a fixed solids loading slurry, the viscosity
rapidly increased with the increase of binder
percent - Maximum solids loading achieved was 55 volume
with 20 volume or less binder in the premix
115
Rheology of the slurry using different slurry
compositions
The green bodies were produced using these slurry
116
Setting of as cast foam
Setting of foams soon after casting is essential
to retain the foam microstructure
Setting of the cast foams was carried out by drop
wise addition of nitric acid
At frequency of 1 Hz and 0.01 strain
Change in viscoelastic behaviour of slurry after
foaming
Gelation behaviour of foams was confirmed by
viscoelastic measurement before and after
addition of acid
117
Viscosity control and its influence on total
porosity
Viscosity of the slurries can be tailored by
binder volume percent in the premix and ceramic
powder loading
Viscosity exerts a strong influence on foaming
behaviour of the slurries and thus total porosity
of the samples
J. Am. Ceram. Soc, under revision
118
Temperature Induced Gelation Behaviour
Temperature induced gelation at 50oC
Viscoelastic property of foamed slurry is
measured at constant frequency of 1 Hz
Measured at constant frequency of 1 Hz and 1
strain
119
References
- Presentation of TA instrument for Rheology of
fluid. - J. Am. Ceram. Soc., 71121062-70 91988).
- Principles of Ceramics Processing- J.S. Reed
- J. Am. Ceram. Soc, 813549-56, 1998. Uematsu
Keizu et. al. - PhD Thesis by S. Dhara
120
A VISCOSITY PRIMER VISCOSITY DEFINITIONS Viscosi
ty Definition A measure of the resistance of
flow due to internal friction when one layer of
fluid is caused to move in relationship to
another layer. The Poise represents absolute
viscosity, the tangential force per unit area of
either of two horizontal planes at unit distance
apart, the space between being filled with the
substance. A liquid with an absolute viscosity of
one Poise requires a force of one dyne to
maintain a velocity differential of one
centimeter per second over a surface one
centimeter square. When the ratio of shearing
stress to the rate of shear is constant, as is
the case with water and thin motor oils, the
fluid is called a Newtonian fluid. In the case of
non-Newtonian fluids, the ratio varies with the
shearing stress, and viscosities of such fluids
are called apparent viscosities. In the new SI
system, it is proposed that values for the Poise
be stated as Pascal seconds, the conversion
factor being 1 Poise equal to 1 10-1 Pas. A
common measurement unit is the milliPascal second
(mPas). Conversion factors are as follows 1
centipoise (cP) 0.01 poise (P) 1 Pas 10 P 1
cP 0.001 Pas 1 mPas 1 Pas 1000 cP
121
Absolute Viscosity Definition The tangential
force per unit area of two parallel planes at
unit distance apart when the space between them
is filled with a fluid and one plane moves with
unit velocity in its own plane relative to the
other. Also known as coefficient of
viscosity. Apparent Viscosity Definition The
value obtained by applying the instrumental
equations used in obtaining the viscosity of a
Newtonian fluid to viscometer measurements of a
non-Newtonian fluid. Dilute Solution
Viscosity Definition The viscosity of a dilute
solution of a polymer, measured under prescribed
conditions, is an indication of the molecular
weight of the polymer and can be used to
calculate the degree of polymerization.
122
Intrinsic Viscosity (?) Definition The ratio
of a solutions specific viscosity to the
concentration of the solute, extrapolated to zero
concentration. Intrinsic viscosity reflects the
capability of a polymer in solution to enhance
the viscosity of the solution. The viscosity
behavior of macromolecular substances in solution
is one of the most frequently used approaches for
characterization. The intrinsic viscosity number
is defined as the limiting value of the specific
viscosity/concentration ratio at zero
concentration. Intrinsic viscosity is determined
by measuring the relative viscosity at several
different concentrations and then extrapolating
the specific viscosity to zero concentration. The
variation of the viscosity number with
concentration depends on the type of molecule as
well as the solvent. In general, the intrinsic
viscosity of linear macromolecular substances is
related to the molecular weight or degree of
polymerization. With linear macromolecules,
viscosity number measurements can provide a
method for the rapid determination of molecular
weight when the relationship between viscosity
and molecular weight has been established.
Intrinsic viscosity is calculated by determining
the reduced viscosity (?sp/c) and extrapolating
to infinite dilution. Huggins equation ?
lim?red c ? 0 Craemer equation ? lim?inh c ? 0