1800s Mendeleev built table in order of increasing atomic mass' - PowerPoint PPT Presentation
1 / 17
Title:
1800s Mendeleev built table in order of increasing atomic mass'
Description:
Most elements are metals and occur on the left side. The nonmetals appear on the right side. ... can these combine to form compounds? Ions & Bonding. Evaluate ... – PowerPoint PPT presentation
Number of Views:86
Avg rating:3.0/5.0
Title: 1800s Mendeleev built table in order of increasing atomic mass'
1
(No Transcript)
2
History
- 1800s Mendeleev built table in order of
increasing atomic mass. - 1913 Moseley revised table to put in order of
increasing atomic number.
3
Rows and Columns
- Periods vs. groups
- 7 periods on the table
- Periods rows energy levels
- Period energy level of the valence electrons
- 18 groups on the table
- Groups columns family valence electrons
- Group A representative elements main group
- Group A of valence electrons
- Group B transition elements
- Group B does NOT reflect the of valence
electrons
4
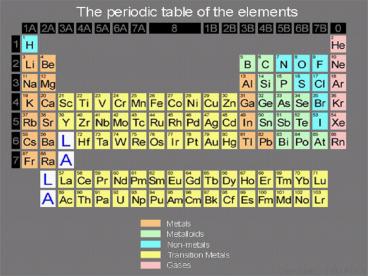
Anatomy
- Most elements are metals and occur on the left
side.
- The nonmetals appear on the right side.
- Metalloids are elements that have some metallic
and some nonmetallic properties.
5
Anatomy
- The periodic table is organized to group elements
with similar properties in vertical columns.
6
(No Transcript)
7
Noble gases elements in which outermost s and p
sublevels are filled Representative elements
outermost s and p sublevel is partially
filled Transition metals outermost s sublevel
and nearly d sublevel contain electrons Inner
transition metals outermost s and nearby f
sublevel contain electrons
Electrons
8
- The octet rule says that atoms will gain, lose or
share electrons in order to achieve a full outer
energy level. This usually means 8 valence
electrons. - Which group on the periodic table already has a
full outer energy level? - All atoms want their electron clouds to look like
noble gases. Atoms strive to become stable by
gaining, losing or sharing as few electrons as
possible. - Consider each Group A column. Will each gain,
lose or share electrons? How many?
Electrons
9
Atomic Radius
What pattern do you notice across a row?
What pattern do you notice up a column?
10
Trend in Atomic Radius
- Radius is...
- Distance from the center of the atom to the
valence electrons.
Period Trend
Group Trend
- WHY?
- Radius decreases across a period because the
nucleus is gaining more protons to become larger
and more strongly positive. No energy levels are
being added. - Radius decreases up a group because there are
fewer energy levels at the top of the periodic
table.
11
Ionization Energy
12
Trend in Ionization Energy
- IE is energy required to remove an electron from
an atom. - Based on what you know about the octet rule,
which atoms might WANT to have electrons removed?
Which ones might NOT WANT to have electrons
removed?
13
Trend in Ionization Energy
- WHY?
- IE increases across a period because the nucleus
is gaining more protons to become larger and more
strongly positive. The nucleus holds tightly to
the electrons. Therefore, HIGH ENERGY is needed
to remove an electron. - IE increases up a group because there are fewer
energy levels at the top of the periodic table.
The pull of the positive nucleus is NOT shielded
by few energy levels. At the bottom of the
table, the pull of the nucleus IS SHIELDED by the
many energy levels. Therefore, LOW ENERGY is
needed to remove an electron from a large atom.
14
Trend in Electron Affinity
- EA is the ability of an atom to attract electrons
to itself. - Think about what you already know...
15
Electronegativity
16
Trend in Electronegativity
Electronegativity is the ability of an atom to
draw electrons from another atom closer to itself
when in a chemical bond.
- WHY?
- EN increases across a period because the nucleus
is gaining more protons to become larger and more
strongly positive. The nucleus holds tightly to
the electrons. Therefore, the strong nucleus is
able to draw the bonding electrons closer. - EN increases up a group because there are fewer
energy levels at the top of the periodic table.
The pull of the positive nucleus is NOT shielded
by few energy levels. At the bottom of the
table, the pull of the nucleus IS SHIELDED by the
many energy levels. Therefore, the shielded
nucleus is unable to draw the bonding electrons
closer.
17
Ions Bonding
- Think about everything that you know...valence
electrons, octet rule, ionization energy,
electron affinity - Who forms positive ions, or cations? How?
- Who forms negative ions, or anions? How?
- How can these combine to form compounds?
18
Evaluate the answer to the question.
- Question Why does IE increase across a period?
- Answer The nucleus gets bigger and stronger.
19
IONIC COMPOUNDS
Ionic compounds are made up of anions and
cations. Remember, cations are metals, and
anions are nonmetals. So....ionic compounds are
made up of A METAL NONMETAL.
20
Covalent Compound
- These compounds differ from ionic compounds.
They are NOT made up of ions. They are
completely composed of NONMETALS.