Daily science- pg. 30 - PowerPoint PPT Presentation
Title:
Daily science- pg. 30
Description:
Daily science- pg. 30 Create a table like the following on page 30 in your notebook. Label this page History of the Atom. Leave a few lines for each scientist. – PowerPoint PPT presentation
Number of Views:16
Avg rating:3.0/5.0
Title: Daily science- pg. 30
1
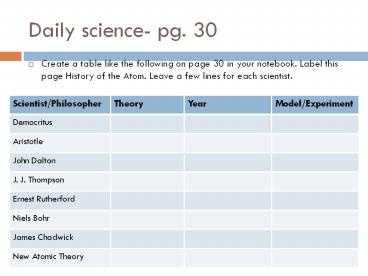
Daily science- pg. 30
- Create a table like the following on page 30 in
your notebook. Label this page History of the
Atom. Leave a few lines for each scientist.
Scientist/Philosopher Theory Year Model/Experiment
Democritus
Aristotle
John Dalton
J. J. Thompson
Ernest Rutherford
Niels Bohr
James Chadwick
New Atomic Theory
2
The Structure of atoms
- Pg. 29
3
What makes up an atom?
- Atom- smallest unit of an element that still
retains that elements properties - Atoms are made of subatomic particles.
- The three subatomic particles are the proton,
electron, and neutron.
4
Structure of an atom
- The nucleus is located at the center of the atom.
- It contains positively charged particles, called
protons, and neutral particles, called neutrons. - Most of the mass is located here.
5
Structure of an atom
- Electrons, or negatively charged particles, exist
outside of the nucleus in the electron cloud.
6
Subatomic Particles chart
Particle Charge Mass Location
Proton 1 1.67 x 10 -27 In the nucleus
Neutron 0 1.67 x 10 -27 In the nucleus
Electron -1 9.11 X 10 -31 Outside the nucleus
7
Atomic Number
- The periodic table can be used to find the number
of protons, electrons, and neutrons. - The atomic number of an element (labeled Z) tells
you how many protons there are. - On the periodic table, the elements are listed as
neutral elements so the number of electronsthe
number of protons.
8
Atomic number
Z
9
Mass Number
- The mass number (labeled A) is the number of
neutrons protons. - Mass number (if neutrons is unknown)atomic
mass rounded to the nearest whole number - The atomic mass is located under the element
symbol. - You can find the number of neutrons by
subtracting the Atomic number (Z) from the Mass
number (A) - A-Z of neutrons
10
Mass Number
11
Writing Atomic number and mass number
- There is a way to represent atomic mass and
number - Steps
- 1-start with the element symbol
- 2- place the mass number on the top left side of
the symbol - 3-place the atomic number on the bottom left side
of the symbol
12
Symbol
Al
27
13
13
Isotopes
- An isotope is an element that has the same number
of protons but a different number of neutrons. - Ex. Carbon has 2 common isotopes
- Carbon-12 Carbon-14
- The numbers represent the mass number
14
Determining the number of neutrons
- Ex. Carbon 12 and Carbon 14
15
Counting Atoms
- Pg. 31
16
Atomic Mass
- Atomic mass units (amu) are used to express an
atoms mass - Atomic mass units are based off the element
Carbon - Found under the element symbol
17
Average atomic mass
- Average atomic mass is a weighted average of the
isotopes of a given element. - More common isotopes have a greater effect on the
average atomic mass than do less common isotopes. - Ex. Chlorine- 37 and Chlorine- 35 (average atomic
mass is 35.45)
18
Percent Abundance
- The atomic mass can be calculated by summing the
products of each isotopes percent abundance and
that isotopes atomic mass. - Mass contribution tells how much of each isotope
contributes to the total atomic mass. - Mass contribution ( abundance) x (atomic mass)
19
Percent Abundance example
- Chlorine 35 has an atomic mass of 34.969 amu and
its percent abundance 75.770. What is its mass
contribution? - Chlorine 37 has an atomic mass of 36.966 amu and
its percent abundance is 24.230. What is its
mass contribution. - Using the two previous answers, what is the
average atomic mass of Chlorine?