Aldehydes from oxidation of primary alcohols using the Dess-Martin periodinane reagent - PowerPoint PPT Presentation
1 / 29
Title:
Aldehydes from oxidation of primary alcohols using the Dess-Martin periodinane reagent
Description:
14.2 Preparing Aldehydes and Ketones Aldehydes from oxidation of primary alcohols using the Dess-Martin periodinane reagent Aldehydes from reduction of carboxylic ... – PowerPoint PPT presentation
Number of Views:99
Avg rating:3.0/5.0
Title: Aldehydes from oxidation of primary alcohols using the Dess-Martin periodinane reagent
1
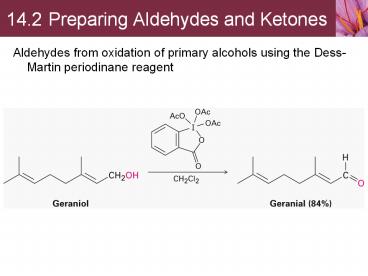
14.2 Preparing Aldehydes and Ketones
- Aldehydes from oxidation of primary alcohols
using the Dess-Martin periodinane reagent
2
Preparing Aldehydes and Ketones
- Aldehydes from reduction of carboxylic esters
using diisobutylaluminum hydride (DIBAH)
3
Preparing Aldehydes and Ketones
- Secondary alcohols are oxidized by variety of
chromium-based reagents to give ketones - Aryl ketones from Friedel-Crafts acylation
reactions
4
Nucleophilic Addition of Grignard and Hydride
Reagents Alcohol Formation
- Mechanism of Grignard Reaction
5
Nucleophilic Addition of Grignard and Hydride
Reagents Alcohol Formation
- In an analogous manner the reaction of aldehydes
and ketones with hydride reagents may be
represented as proceeding through a nucleophilic
addition of a hydride ion (H) to the CO carbon - LiAlH4 and NaBH4 act as if they are donors of
hydride ion
6
14.7 Nucleophilic Addition of Amines Imine and
Enamine Formation
- Primary amines, RNH2, add to aldehydes and
ketones to yield imines, R2CNR - Secondary amines, R2NH, add similarly to yield
enamines, R2N-CRCR2
7
Nucleophilic Addition of Amines Imine and
Enamine Formation
- Imines are common biological intermediates where
they are often called Schiff bases
8
Nucleophilic Addition of Amines Imine and
Enamine Formation
- Imine and enamine formations reach maximum rate
around pH 4 to 5 - Slow at pH gt 5 because there is insufficient H
present in solution to protonate intermediate
carbinolamine OH to yield the better leaving
group OH2 - Slow at pH lt 4 because the basic amine
nucleophile is protonated and initial
nucleophilic addition cannot occur
9
Worked Example 14.1Predicting the Product of
Reaction between a Ketone and an Amine
10
Nucleophilic Addition of Alcohols Acetal
Formation
- Acetal and hemiacetal groups are common in
carbohydrate chemistry - Glucose, a polyhydroxy aldehyde, undergoes
intramolecular nucleophilic addition - Exists primarily as a cyclic hemiacetal
11
14.9 Nucleophilic Addition of Phosphorus Ylides
The Wittig Reaction
- Wittig reaction
- Converts aldehydes and ketones into alkenes
- Phosphorus ylide, R2CP(C6H5)3, adds to aldehyde
or ketone to yield dipolar, alkoxide ion
intermediate - Ylide (pronounced ill-id) is a neutral, dipolar
compound with adjacent positive and negative
charges - Also called a phosphorane and written in the
resonance form R2CP(C6H5)3 - Dipolar intermediate spontaneously decomposes
through a four-membered ring to yield alkene and
triphenylphosphine oxide, (Ph)3PO - Wittig reaction results in replacement of
carbonyl oxygen with R2C group of original
phosphorane
12
Nucleophilic Addition of Phosphorus Ylides The
Wittig Reaction
Wittig reaction mechanism
13
Nucleophilic Addition of Phosphorus Ylides The
Wittig Reaction
- Phosphorus ylides are prepared by SN2 reaction of
primary and some secondary alkyl halides with
triphenylphosphine, (Ph)3P, followed by treatment
with base
14
Nucleophilic Addition of Phosphorus Ylides The
Wittig Reaction
- Wittig reactions used commercially to synthesize
numerous pharmaceuticals
15
Worked Example 14.3Synthesizing an Alkene
Using a Wittig Reaction
- What carbonyl compound and what phosphorus ylide
might you use to prepare 3-ethylpent-2-ene?
16
Worked Example 14.3Synthesizing an Alkene
Using a Wittig Reaction
Solution
17
Biological Reductions
- Cannizzaro reaction is a nucleophilic acyl
substitution reaction of aldehydes and ketones - OH adds to aldehyde to give tetrahedral
intermediate - H ion is transferred to a second aldehyde
- The aldehyde accepting the H ion is reduced and
the aldehyde transferring the H is oxidized
18
Biological Reductions
- Cannizzaro reaction mechanism is analogous to
biological reduction in living organisms by
nicotinamide adenine dinucleotide, NADH - NADH donates H to aldehydes and ketones,
similar to tetrahedral alkoxide intermediate in
Cannizzaro reaction
19
Conjugate Nucleophilic Addition to
a,ß-Unsaturated Aldehydes and Ketones
- Conjugate addition occurs because the nucleophile
can add to either one of two electrophilic
carbons of the a,b-unsaturated aldehyde or ketone
20
Conjugate Nucleophilic Addition to
a,ß-Unsaturated Aldehydes and Ketones
- Conjugated double bond of a,b-unsaturated
carbonyl is activated by carbonyl group of the
aldehyde or ketone - CC double bond is not activated for addition in
absence of carbonyl group
21
Conjugate Nucleophilic Addition to
a,ß-Unsaturated Aldehydes and Ketones
- Primary and secondary amines add to
a,b-unsaturated aldehydes and ketones to yield
b-amino aldehydes and ketones - Both 1,2- and 1,4-addition occur
- Additions are reversible
- More stable conjugate addition product
accumulates
22
Conjugate Nucleophilic Addition to
a,ß-Unsaturated Aldehydes and Ketones
- Conjugate addition of an alkyl or other organic
group to an a,b-unsaturated ketone (but not
aldehyde) is a useful 1,4-addition reaction
23
Conjugate Nucleophilic Addition to
a,ß-Unsaturated Aldehydes and Ketones
- Conjugate addition of alkyl groups to an
a,b-unsaturated ketone (not aldehyde) is
accomplished with a lithium diorganocopper
reagent, R2CuLi (Gilman reagent) - Lithium diorganocopper reagent is prepared by
reaction of 1 equivalent of copper(I) iodide and
2 equivalents of an organolithium reagent, RLi - Organolithium reagent is prepared by reaction of
lithium metal with an organohalide
24
Conjugate Nucleophilic Addition to
a,ß-Unsaturated Aldehydes and Ketones
- Primary, secondary, and even tertiary alkyl
groups undergo conjugate addition - Alkynyl groups react poorly
- Grignard reagents and organolithium reagents
normally give direct carbonyl addition to
a,b-unsaturated ketones
25
Worked Example 14.4Using a Conjugate Addition
Reaction
- How might you use a conjugate addition reaction
to prepare 2-methyl-3-propylcyclopentanone?
26
Worked Example 14.4Using a Conjugate Addition
Reaction
- Solution
27
Spectroscopy of Aldehydes and Ketones
28
Spectroscopy of Aldehydes and Ketones
- Aldehyde protons (RCHO) absorb near 10 d in the
1H NMR - Aldehyde proton shows spin-spin coupling with
protons on the neighboring carbon, with coupling
constant J 3 Hz - Hydrogens on carbon next to a carbonyl group are
slightly deshielded and absorb near to 2.0 to 2.3
d
29
Spectroscopy of Aldehydes and Ketones
- Carbonyl-group carbon atoms of aldehydes and
ketones have characteristic 13C NMR resonances in
the range of 190 to 215 d