Chapter 12: Alkenes and Alkynes - PowerPoint PPT Presentation
1 / 22
Title:
Chapter 12: Alkenes and Alkynes
Description:
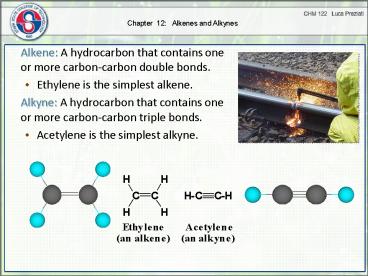
Chapter 12: Alkenes and Alkynes Alkene: A hydrocarbon that contains one or more carbon-carbon double bonds. Ethylene is the simplest alkene. Alkyne: A hydrocarbon ... – PowerPoint PPT presentation
Number of Views:227
Avg rating:3.0/5.0
Title: Chapter 12: Alkenes and Alkynes
1
Chapter 12 Alkenes and Alkynes
- Alkene A hydrocarbon that contains one or more
carbon-carbon double bonds. - Ethylene is the simplest alkene.
- Alkyne A hydrocarbon that contains one or more
carbon-carbon triple bonds. - Acetylene is the simplest alkyne.
2
Chapter 12 Alkenes and Alkynes
- Cis-trans isomerism Double Bond No Free
rotation!!! - Because of restricted rotation about a
carbon-carbon double bond, an alkene with two
different groups on each carbon of the double
bond shows cis-trans isomerism.
3
Chapter 12 Alkenes and Alkynes
- To name an alkene
- The parent name is that of the longest chain that
contains the CC. - Number the chain from the end that gives the
lower numbers to the carbons of the CC. - Locate the CC by the number of its first carbon.
- Use the ending -ene to show the presence of the
CC - Branched-chain alkenes are named in a manner
similar to alkanes in which substituted groups
are located and named.
4
Chapter 12 Alkenes and Alkynes
- To name an alkyne
- Follow the same rules as for alkenes, but use the
ending -yne to show the presence of the triple
bond.
Common names are still used for some alkenes and
alkynes, particularly those with low molecular
weight.
5
Chapter 12 Alkenes and Alkynes
- To name a cycloalkene
- Number the carbon atoms of the ring double bond 1
and 2 in the direction that gives the lower
number to the substituent encountered first. - Note that it is not necessary to explicitly
number the position of the double bond in a
cycloalkene as in linear alkenes. - Number and list substituents in alphabetical
order.
6
Chapter 12 Alkenes and Alkynes
- Alkenes that contain more than one double bond
are named as alkadienes, alkatrienes, and so
forth. - Those that contain several double bonds are
referred to more generally as polyenes (Greek
poly, many).
Alkenes and alkynes physical properties are very
similar to the alkanes properties
7
Chapter 12 Alkenes and Alkynes
- Terpene A compound whose carbon skeleton can be
divided into five-carbon units identical with the
carbon skeleton of isoprene. - Terpenes illustrate an important principle of the
molecular logic of living systems. - In building large molecules, small subunits are
bonded together by a series of enzyme-catalyzed
reactions and then chemically modified by
additional enzyme-catalyzed reactions.
8
Chapter 12 Terpenes
9
Chapter 12 Alkenes and Alkynes
The most common reaction is addition to the
double bond.
10
Chapter 12 Reactions
- Combustion (but we already spoke about it in
chp.11) - Addition to the double bond.
- Polymerization
11
Chapter 12 Alkenes and Alkynes
- Most alkene addition reactions are exothermic.
- the products are more stable (lower in energy)
than the reactants. - Just because they are exothermic doesnt mean
that alkene addition reactions occur rapidly. - Reaction rate depends on the activation energy.
- Many alkene addition reactions require a catalyst.
12
HX
- Addition of HX (HCl, HBr, or HI) to an alkene
gives a haloalkane. - H adds to one carbon of the CC and X to the
other. - Reaction is regioselective. One direction of bond
forming (or bond breaking) occurs in preference
to all other directions. - Markovnikovs rule H adds to the less
substituted carbon and X to the more substituted
carbon.
13
HX
- Chemists account for the addition of HX to an
alkene by a two-step reaction mechanism. - We use curved arrows to show the repositioning of
electron pairs during a chemical reaction. - The tail of an arrow shows the origin of the
electron pair (either on an atom or in the double
bond). - The head of the arrow shows its new position.
- Curved arrows show us which bonds break and which
new ones form.
14
HX
- Step 1
- Reaction of the carbon-carbon double bond with H
gives a secondary (2) carbocation intermediate,
a species containing a carbon atom with only
three bonds to it and bearing a positive charge. - Step 2
- Reaction of the carbocation intermediate with
chloride ion completes the addition.
15
H20
- Addition of water is called hydration.
- Hydration is acid catalyzed, most commonly by
H2SO4. - Hydration follows Markovnikovs rule H adds to
the less substituted carbon and OH adds to the
more substituted carbon.
16
H20
- Step 1
- Step 2
- Step 3
17
Cl2 / Br2
- Addition takes place readily at room temp.
- Reaction is generally carried out using pure
reagents, or mixing them in a nonreactive organic
solvent. - Addition of Br2 is a useful qualitative test for
the presence of a carbon-carbon double bond. - Br2 has a deep red color dibromoalkanes are
colorless.
18
H2
- Virtually all alkenes add H2 in the presence of
a transition metal catalyst, commonly Pd, Pt, or
Ni.
19
H2
?
lt Wilhelm Normann invented what he called fat
hardening. Yoday known as Hydrogenation
The process of hydrogenation adds hydrogen
atoms to cis-unsaturated fats, eliminating double
bonds and making them into partially or
completely saturated fats. However, partial
hydrogenation converts a part of cis-isomers into
trans-unsaturated fats instead of hydrogenating
them completely .
Coronary heart disease
http//en.wikipedia.org/wiki/Trans_fat
20
Polymerization
- From the perspective of the organic chemical
industry, the single most important reaction of
alkenes is polymerization - polymer Greek poly, many, and meros, part any
long-chain molecule synthesized by bonding
together many single parts, called monomers. - monomer Greek mono, single and meros, part.
polypropene (polypropylene)
21
Polymerization
- Low-density polyethylene (LDPE)
- A highly branched polymer polymer chains do not
pack well and London dispersion forces between
them are weak. - Softens and melts above 115C.
- Approximately 65 of all LDPE is used for the
production of films for packaging and for trash
bags. - High-density polyethylene (HDPE)
- Only minimal chain branching chains pack well
and London dispersion forces between them are
strong. - Has higher melting point than LDPE and is
stronger - Can be blow molded to squeezable jugs and
bottles.
22
Polymerization